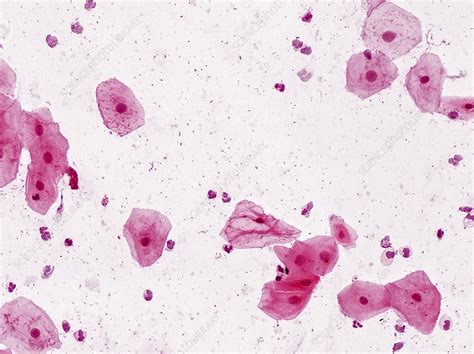
Interpreting cytology results, particularly those involving squamous epithelial cells, demands precision and a nuanced understanding of cellular patterns, staining techniques, and clinical correlations. The results labeled as "610" often refer to a specific diagnostic code or categorization within cytology reports, guiding clinicians and pathologists toward possible diagnoses. Yet, misinterpretation of these results can lead to unnecessary anxiety, inappropriate treatments, or overlooked malignancies. Thus, a meticulous approach rooted in expert knowledge is essential to navigate the common pitfalls associated with squamous epithelial cell analysis.
Understanding the Significance of Squamous Epithelial Cells in Cytology

Squamous epithelial cells line a variety of mucosal surfaces, including the cervix, oral cavity, and skin. Their appearance in cytological samples—such as Pap smears—serves as a critical screening tool for premalignant and malignant processes, especially in cervical pathology. The categorization of squamous cells, including abnormal features, dysplasia, or benign keratinization, informs diagnostic pathways and subsequent management. The “610” result, within this context, may indicate a specific observation or classification, such as a particular abnormality pattern, or it may correspond to a numerical code used in reporting software. However, interpreting these results without understanding their broader context fosters the risk of errors.
What Does the “610” Result Typically Signify?
To grasp the potential pitfalls, clinicians and cytotechnologists must clarify what “610” refers to within their specific reporting domain. Often, laboratory coding systems or standardized schemas like the The Bethesda System for Reporting Cervical Cytology may assign numerical codes for certain cellular findings or categories. For example, a code like 610 could correspond to a diagnosis such as low-grade squamous intraepithelial lesion (LSIL), atypical squamous cells of undetermined significance (ASC-US), or another specific category. Misconceptions arise when interpreting such codes without cross-referencing the exact definitions provided by the reporting system or understanding the morphological criteria that underpin them.
| Relevant Category | Substantive Data |
|---|---|
| Common Interpretation | Potentially encompasses benign reactive changes, infections, or dysplastic alterations |
| Prevalence | Up to 5% of routine Pap tests, with varying implications based on age and risk factors |

Common Mistakes in Interpreting “610” Squamous Cell Results

Errors in interpretation typically stem from a combination of over-reliance on numeric coding, insufficient morphological analysis, and neglecting patient-specific factors. Recognizing these pitfalls guides clinicians in refining their diagnostic acumen and ensuring patient safety.
1. Equating Numeric Codes with Definitive Diagnoses
One of the most pervasive mistakes is assuming that a code like 610 inherently confirms a particular pathology. Numeric identifiers are merely abbreviations within a reporting schema—they do not replace comprehensive morphological assessment. For example, mistaking a “610” code for benign or malignant status without reviewing cell features such as nuclear pleomorphism, keratinization, or nuclear-cytoplasmic ratios risks misclassification.
2. Overlooking Cellular Morphology and Context
Squamous cells can appear abnormal due to reactive processes, inflammation, or technical artifacts. Failing to distinguish these from true dysplastic changes represents a key interpretive error. For example, reactive atypia caused by inflammation may mimic low-grade lesions, leading to false positives if not carefully evaluated.
3. Ignoring Ancillary Testing and Clinical Correlation
In the era of integrated diagnostics, relying solely on cytology results—especially codes like “610”—without correlating with HPV testing, colposcopic findings, or patient risk factors increases misclassification risk. For instance, a low-grade cytological abnormality may warrant watchful waiting if HPV status suggests low risk, versus aggressive intervention if high-risk HPV types are detected.
4. Misinterpretation Due to Technical Artifacts
Sample preparation artifacts such as crushing, overlapping cells, or stain artifact can distort cellular morphology. Overlooking such errors leads to false interpretations. Recognizing artifact characteristics—such as irregular nuclear borders or cytoplasmic smudging—is critical to avoid misdiagnosis.
Strategies to Improve Accuracy in Squamous Cell Interpretation
Expert practitioners follow a set of disciplined strategies to circumvent common mistakes. These include rigorous morphological analysis, contextual biological understanding, and utilization of confirmation tools. Implementing these practices enhances diagnostic confidence and patient outcomes.
1. Detailed Morphological Analysis
Focus on nuclear features—size, shape, chromatin pattern, membrane integrity—as well as cytoplasmic details. Compare findings against established criteria for benign, reactive, and dysplastic cells. Pattern recognition plays an essential role, especially in differentiating between hyperkeratosis, koilocytosis, and true dysplasia.
2. Cross-Referencing Clinical Data and Ancillary Tests
Incorporate HPV DNA testing results, patient age, immune status, and previous cytology history. A composite assessment reduces overdiagnosis or underdiagnosis and guides appropriate management.
3. Recognizing Artifacts and Sampling Limitations
Become adept at differentiating true cellular abnormalities from technical distortions. Adequate sample collection, proper fixation, and staining quality control are fundamental. When in doubt, repeat sampling or adjunct testing can clarify ambiguous findings.
Final Thoughts: Navigating the Complexities of Squamous Cell Results
The interpretation of “610” or any such coded result associated with squamous epithelial cells requires a multifaceted approach—combining morphological expertise, clinical judgment, and laboratory science. Precision in delineating benign from potentially malignant lesions hinges on continual awareness of common pitfalls and a commitment to analytical rigor. By cultivating these competencies, clinicians and cytopathologists can mitigate misinterpretations, thereby safeguarding patient health and improving diagnostic accuracy.